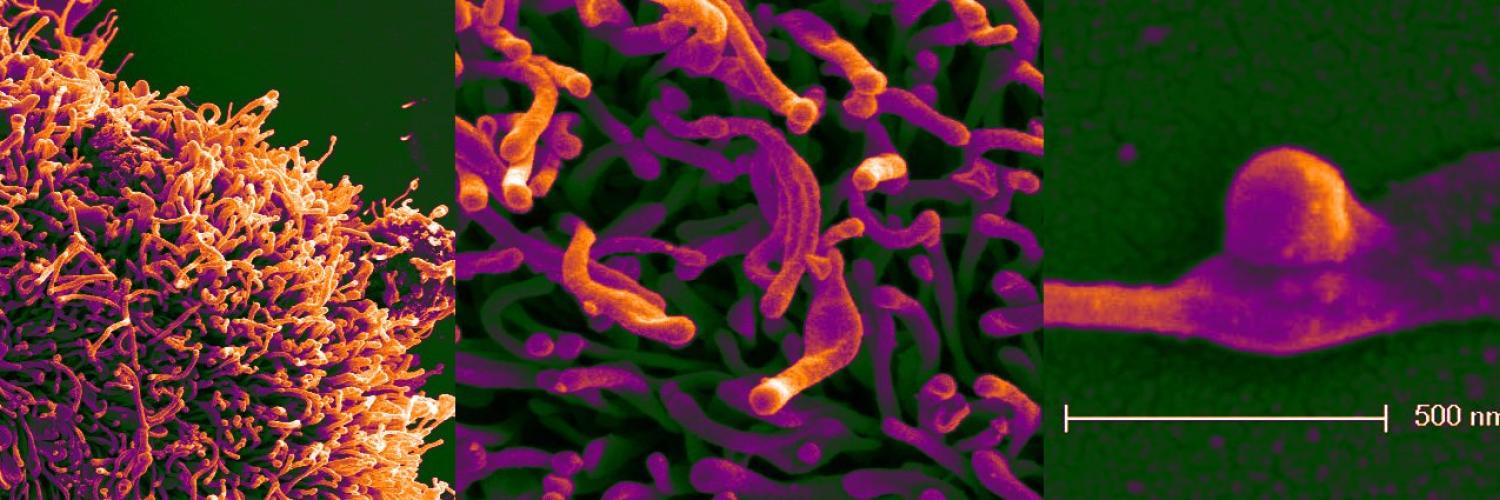
Cells have many self-regulating processes that create the optimal internal state for them to survive and thrive: 'homeostasis' enables cells to respond to external changes and governs their capacity to synthesise new molecules.
The homeostatic state of the cell when it encounters pathogens therefore determines the success or failure of the infection and depends on factors like the time of day, development stage, prior cellular activity and physiological stress.
We're exploring how mechanisms of host cell osmotic and protein homeostasis impact susceptibility to infection, immune responses and the
production of new virus particles for transmission to others. For example, individual cells have molecular circadian clocks that generate daily rhythms in protein metabolism that alters their homeostatic balance over 24 hours.
Viruses must co-opt fundamental cellular functions like protein synthesis to replicate, and we find that the amount of virus produced by cells and disease severity in animals depends on the time that cells are infected. We use physiologically relevant primary cells and natural host-pathogen models to test our hypotheses. In addition, we apply our fundamental research to find novel ways to prevent virus transmission, focusing on respiratory infections such as SARS-CoV-2 and influenza.