The first drug to treat HIV was approved by the FDA in 1987, just four years after HIV had been identified as the agent behind the AIDS crisis. Despite major progress in controlling HIV with lifelong antiretroviral therapy, some enduring biological mysteries have prevented scientists from finding a cure. Viral resistance to existing drugs also remains a significant challenge, underscoring the need for new therapeutic approaches.
“HIV is one of the most intensively studied viruses. It’s remarkable that we are still uncovering new aspects of its anatomy. Discoveries like this help us move closer to new treatment strategies and raise exciting new questions for future research.”
Principal Group Leader
Virologist Peter Cherepanov has been studying these biological mysteries, specifically the role of a key HIV enzyme called integrase, and now his team have answered a question that followed him for much of his career.
“Integrase is the enzyme HIV uses to insert the viral DNA into the host chromosomes. It’s one of the reasons why HIV cannot yet be cured, because once inserted, the viral DNA can persist permanently in infected cells,” says Peter.
Since the 1990s, studies hinted that integrase might play another role in the HIV life cycle, beyond its well-known function of DNA integration. “If you genetically alter integrase, you often find that viral particles fail to properly form,” Peter explains. “The formation of infectious virus happens before the viral DNA is produced and depends on integrase binding to viral RNA genome.”
Form and function
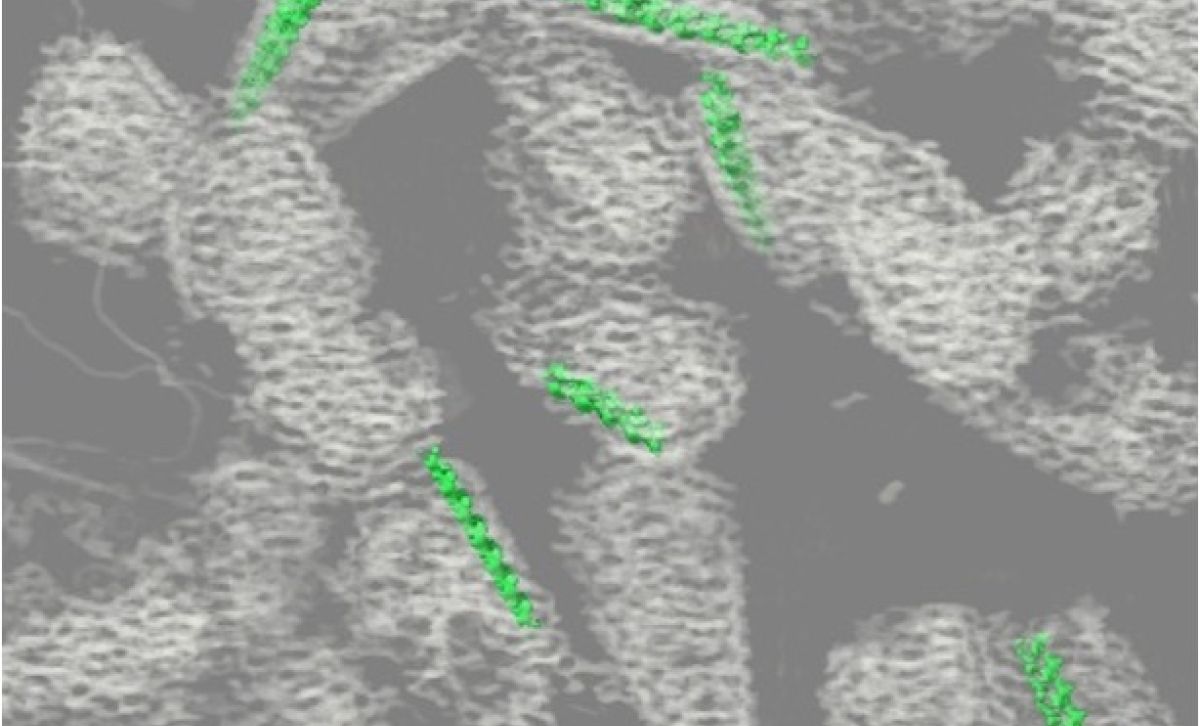
In new research published today in Nature, Peter and his team teamed up with the Crick’s Structural Biology and Proteomics facilities to reveal the first structure of integrase bound to RNA.
“What we saw was astonishing; integrase together with RNA formed long filaments that could be overlayed almost perfectly over the structure of another part of the virus, the capsid,” says Peter. The capsid acts as a protective shell that transports HIV’s genetic material and shields it from cellular defences.
“This was an exciting time; we were 3D printing integrase and capsid structures trying to visualise how they might bind. We obtained a sample of native HIV capsids from colleagues in Peijun Zhang’s laboratory at Oxford University, and when we examined them using our electron microscopes, we found that these integrase filaments indeed lined the inner walls of the capsids.”
“We couldn’t have predicted this using current AI tools. It’s a weak interaction, but integrase and capsid appear to fasten together like Velcro,” Peter explains.
The scientists then teamed up with virologist Alan Engelman from Harvard University and computational chemist Juan Perilla from the University of Delaware, to validate these findings.
“Together, we found that integrase was anchoring the RNA to the inner surface of the HIV capsid, securing it for transport,” Peter explains. “This means that integrase has two distinct roles depending on whether it’s dealing with the viral RNA or DNA, like having two sides to its personality.”
HIV encodes only 15 proteins, so the virus must use them efficiently. “Integrase isn’t just waiting for DNA to appear later in infection, it’s already working much earlier in the viral life cycle,” Peter adds.
Treatment potential
Current HIV therapies include strand transfer inhibitors that block the enzymatic function of integrase. The newly determined structures reveal opportunities to develop drugs that directly interfere with integrase’s interaction with the capsid. The team predict that inhibitors targeting sites close to where integrase interfaces with the inner wall of the capsid, may be most effective at blocking the enzyme’s second role.
They now want to understand how integrase supports reverse transcription, how it transitions between its two roles, and how it ultimately disengages from the viral core.
“HIV is one of the most intensively studied viruses,” Peter concludes. “It’s remarkable that we are still uncovering new aspects of its anatomy. Discoveries like this help us move closer to new treatment strategies and raise exciting new questions for future research.”
This study was funded by The Wellcome Trust, Medical Research Council, Cancer Research UK, US National Institutes of Health, US National Science Foundation